Ozone: the good, the bad and the ugly?
A highly reactive gas plays different roles in different parts of the atmosphere
How can a simple molecule be “good” for us in one place and “bad” in another. There is an adage among some gardeners suggesting that weeds are just plants in the wrong place. Thoreau, termed his garden of beans at Walden Pond a weed he had sown and cultivated at the expense of others. From Emerson to Michael Pollan, weed versus plant has been a rich theme in gardening literature.
Our topic here is ozone, and ozone is that molecule and it is indeed “good” in one place and “bad” in another, to be encouraged or diminished by its impact on each of us. Calling it a “weedy” gas might not get us very far, but you get the idea.
So what is ozone and how can it be both and good and bad. We will get to the ugly part a little later, and as beauty is in the eye of beholder, you can judge for yourself. Time to rein in all these analogies and metaphors and talk some science.
Ozone is a highly reactive form of the second most common gas in the atmosphere. At 21%, “regular” oxygen is comprised of two atoms of this element linked in a relatively stable double bond (O2) It is second in abundance only to the exceptionally stable, triple bonded nitrogen (78% of our atmosphere). Such high concentrations of these two gases are evidence of Life on Earth that would be visible to any outer space observers. J.E. Lovelock in the original Gaia, defined life at the planetary level as the use of the sun’s energy to maintain chemical disequilibrium.
And the presence of free oxygen in our atmosphere is a primary indicator of that disequilibrium. It is the waste product of photosynthesis, and is maintained at that high concentration in our atmosphere by the global vigor of that biological process. Respiration, the counterpoint to photosynthesis, is the use of that oxygen to power large, mobile animals, like us, and most other living things, through oxidation of the products of photosynthesis. Without green plants and photosynthesis, oxygen would be gone from our atmosphere in a geological heartbeat. Disequilibrium as a result of biology – Life.
Ozone adds a third atom of oxygen to a molecule of regular oxygen, is written as O3, and becomes MUCH more reactive! It takes the most intense forms of sunlight (along with other elements) to create ozone. Being so reactive, ozone breaks down rapidly under most conditions, and so it’s presence requires continuous production. While O2 is with us at 21% of the air we breathe, O3 is measured in parts per billion.
Ozone has had a rich history of discovery and application, much of which is captured on the relevant Wikipedia pages ( linked below). Let’s concentrate here on two manifestations of the good and the bad.
The “good” ozone became something a media as well as a science star in the 1970s, and represents one of the fastest global policy responses to an environmental issue.
Ozone is one of the best of atmospheric gases at absorbing high-energy ultraviolet (UV) radiation. It is also created by that same radiation. In a region of the upper atmosphere, at a height of about 15 miles, intense UV radiation from the sun can split O2 into two separate atoms. When one combines with a molecule of O2, ozone (O3) is formed. This reaction, and the slightly elevated concentrations of ozone at this height, have given rise to the term “ozone layer.”
So UV radiation drives the formation of ozone, but it is those same ozone molecules that then absorb additional UV radiation, protecting life at the surface.
But as a highly reactive gas, ozone can break down again rapidly into O2. How rapidly ozone is both formed and destroyed is affected by concentrations of rare forms of other elements. Illuminating the complexity of these reactions won the Nobel Prize for three Scientists (Paul Crutzen, Mario Molina and F. Sherwood Rowland).
One of the elements that can speed the break down of ozone is chlorine – and here is where ozone, and a common industrial compound, became media stars.
Chlorofluorocarbons (CFCs) were first created in the 1920s, and after World War II became widely used in air conditioners and refrigerators, as well as a propellent in spray cans. Non-toxic and stable at ground-level, worldwide adoption led to production of more than a million tons per year.
What Molina et al. found in uncovering the chemical mysteries of ozone and CFCs, was that the same high-energy environment in which ozone is produced can also lead to the destabilization of CFCs and the release of chlorine atoms, which in turn could disrupt the cycle of ozone creation and break down, reducing the concentration of this good shield.
When ozone monitoring stations, and then satellite-based measurements, confirmed significant losses of ozone, especially over Antarctica, the “Ozone Hole” controversy was ignited. The Hole over Antarctica was the poster child for this issue as the extended period of darkness in winter reduced the rate of formation of ozone (which requires sunlight) and allowed more time for the chlorine to work its destructive power.
I use this in classes as an example of an environmental success story, albeit an incomplete one.
Given an understanding of the chemistry of ozone formation and depletion, a global conclave in Montreal yielded a Protocol by the same name which proposed a complete ban on the production of CFCs globally beginning in 1989. The U.S. had acted even more quickly, banning the use of CFCs in aerosol cans in 1978.
While vigilance is required in monitoring the production of CFCs, as shown by a recent increase in rogue production in China, the concentrations of CFCs in the upper atmosphere is decreasing slowly as is the annual size of the ozone hole. This will take a while.
So now for the “Bad” ozone.
Most of us have experienced this directly. Ozone is a major constituent of urban air pollution known colloquially as “smog.” That will get us to the “ugly” part in a minute.
This is the same molecule (O3) that protects us from high energy UV radiation, but in this case it is the elevated presence in the air that breathe, down here, that is the cause for concern.
Ozone is especially reactive with soft tissues like lungs and the internal spaces in plant leaves. It essentially oxidizes membranes, causing damage that is either metabolically expensive to repair, or permanent. The effect of ozone can present as an accelerated aging process. In tree leaves, for example, the rate of carbon gain through photosynthesis is permanently reduced by cumulative ozone exposure, mimicking and accelerating a decline that occurs naturally over a leaf’s lifespan. Lungs can be prematurely aged as well.
The chemistry of ozone formation at ground level is also complex, but is driven by increased concentrations of two by-products of inefficient combustion, oxides of nitrogen and carbon. Carbon in this case is not carbon dioxide, released when combustion of fuels is “complete” but rather forms (in a category called volatile organic carbons, or VOCs) that are only partially combusted or just evaporated directly (volatilized) from gasoline and other products. Formation of ground level ozone also requires sunlight.
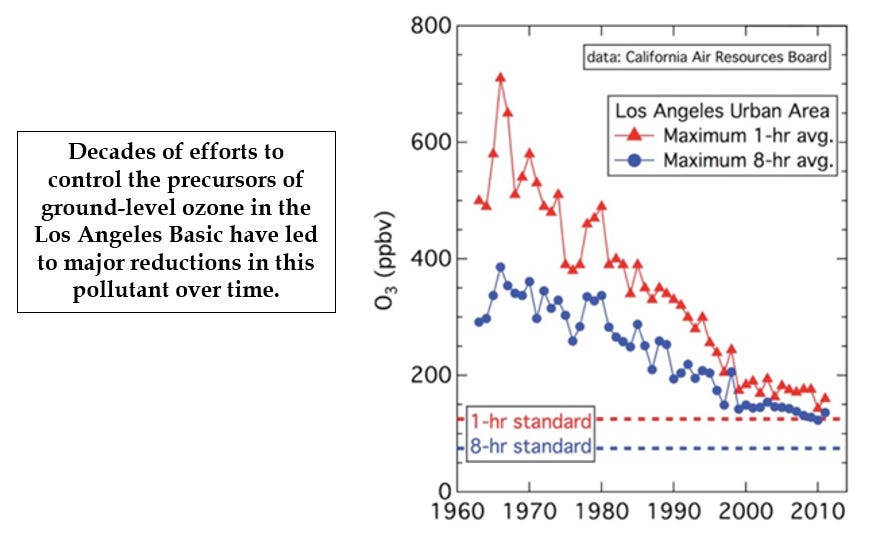
California was the first jurisdiction to look seriously at the health hazards of ozone, and Los Angeles was ground zero for the concern. The LA basin combines large numbers of cars with bright sunshine in a region surrounded by high mountains that can limit the outflow of pollutants. During times of “inversions” – where the air in the basin is trapped for days at a time, ozone concentrations can reach dangerous levels.
In the 1950s it was not unusual for “smog alerts” to require that school children stay indoors and that outside play or any physical activity be limited.
This is another environmental success story that I like to use in class.
As with ozone in the upper atmosphere, ground-level ozone is also highly reactive, and with objects other than lungs and leaves, and so disappears rapidly at night under most circumstances. It needs to be regenerated each day. Hence the negative impacts of ground-level ozone are monitored in terms of both peak daytime concentration (1 hour average) as well as an 8 hour average.
The health effects of ozone were recognized early on, and once the chemistry behind its formation was uncovered, steps could be taken to reduce the cause. California is the poster child here, where regulation to reduce the concentrations of partially oxidized forms of nitrogen and carbon in the atmosphere took the form of improvements in engineering automobile exhausts, as well as other measures.
The success story here is the long-term reduction, by as much as 80%, of those incomplete products of combustion, and in ozone levels as well.
So a single, simple molecule, with the same physical and chemical characteristics either at ground-level or 15 miles up, plays two very different environmental roles. The “good” ozone in the upper atmosphere, produced by high-energy UV radiation, protects us all down here from the harmful effects of that same radiation. The “bad” ozone at ground level attacks lungs and plants (and other things) shortening life spans and reducing biological function.
In both cases, we have learned what causes ozone to be good or bad, and have taken steps to mitigate the negative impacts.
So what about the “ugly?” As pointed out at the top of the essay – in the eye of the beholder. Ozone absorbs visible blue light as well as well as UV, but less at the red end of the spectrum. When present in elevated concentrations, this selective absorbance produces some of those vivid, red/orange summer sunsets.
Beautiful to see, but, knowing the cause, perhaps a bit ugly as well?
Sources
Many others have used the Good/Bad/Ugly context for ozone. A web search will find them!
Here is a good slide presentation from a NASA scientist:
https://www.google.com/search?client=firefox-b-1-d&q=ozonw+goos+bad+ugly
Michael Pollan has penned a brilliant dissection of the weed/plant dichotomy, drawing on Emerson, Thoreau, and his own experience:
https://michaelpollan.com/articles-archive/weeds-are-us/
Ozone Hole
Wikipedia pages include:
https://en.wikipedia.org/wiki/Ozone_depletion#Research_history
https://en.wikipedia.org/wiki/Montreal_Protocol
https://en.wikipedia.org/wiki/Ozone_layer
https://en.wikipedia.org/wiki/Chlorofluorocarbon
The Nobel Prize announcement is here:
https://www.nobelprize.org/prizes/chemistry/1995/summary/
Data and graphics on the ozone hole can be found here:
https://ozonewatch.gsfc.nasa.gov/
https://ozonewatch.gsfc.nasa.gov/facts/SH.html
Graph on change in CFCs over time is from:
https://gml.noaa.gov/hats/graphs/graphs.html
An article on the discovery of “rogue” emissions of CFCs from China is here:
https://www.nature.com/articles/d41586-019-02109-2
Ozone as Smog
Background information on ozone as smog can be found here:
http://photobiology.info/Hockberger.html
https://en.wikipedia.org/wiki/Ozone#History
The graphic on the formation of ground-level ozone is from:
https://www.nsf.gov/news/mmg/mmg_disp.jsp?med_id=76600&from=
The graph on reduction in ozone levels in the LA basin can be found here:
https://eos.org/features/urbanization-air-pollution-now
A history of steps taken to reduce air pollution in California is here:
https://ww2.arb.ca.gov/resources/documents/history-californias-ambient-air-quality-standards
The image of a red summer sunset is from: